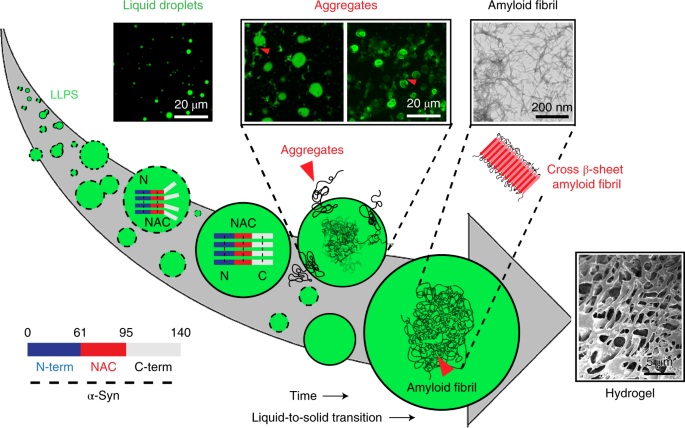
Structure, function and toxicity of alpha‐synuclein: the Bermuda triangle in synucleinopathies - Villar‐Piqué - 2016 - Journal of Neurochemistry - Wiley Online Library
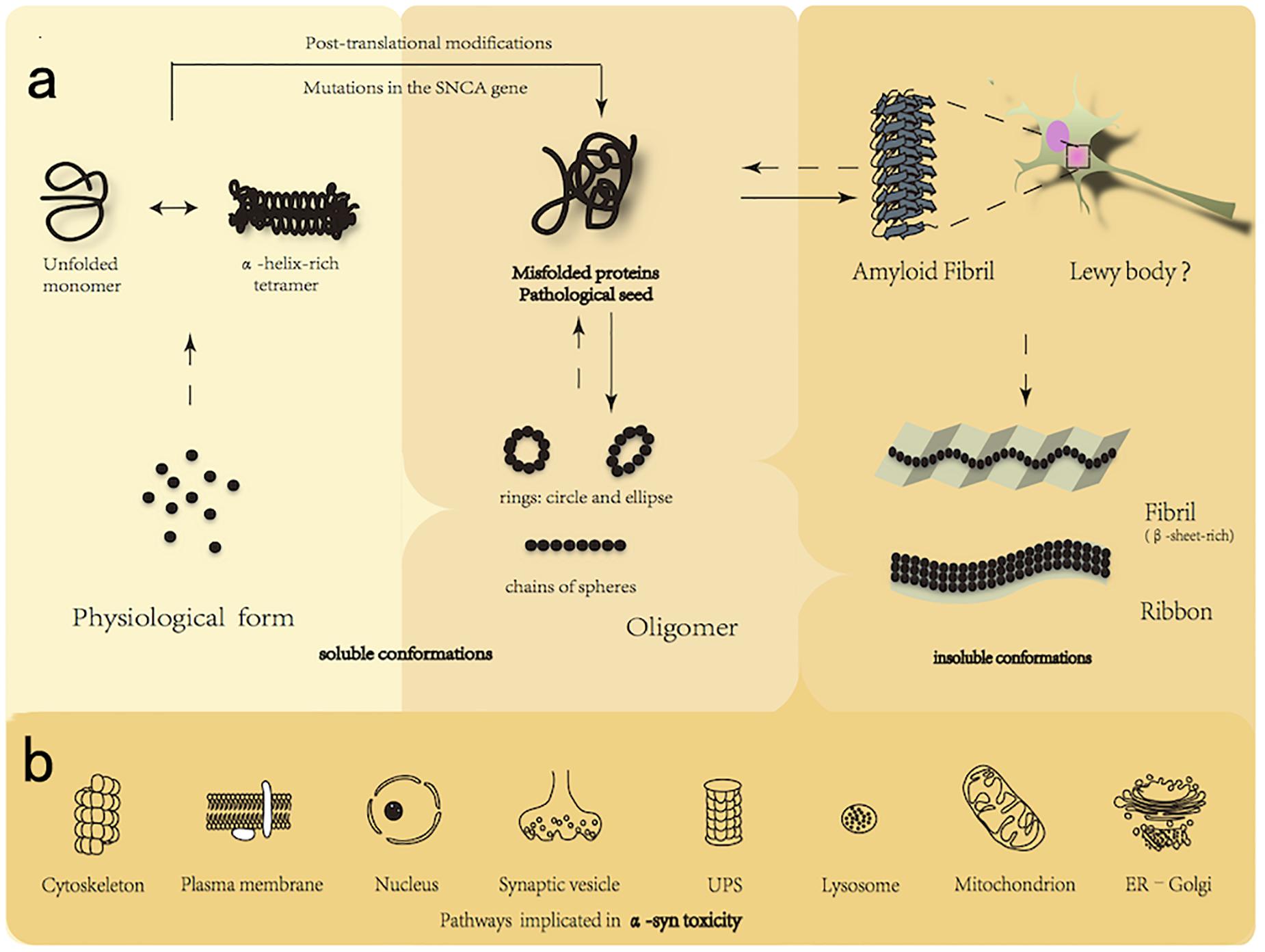
Frontiers | New Perspectives on Roles of Alpha-Synuclein in Parkinson's Disease | Frontiers in Aging Neuroscience
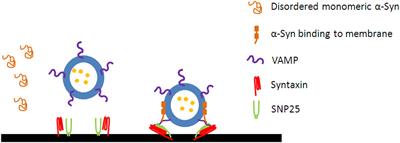
Frontiers | α-Synuclein: A Multifunctional Player in Exocytosis, Endocytosis, and Vesicle Recycling | Neuroscience

α-Synuclein Aggregates Increase the Conductance of Substantia Nigra Dopamine Neurons, an Effect Partly Reversed by the KATP Channel Inhibitor Glibenclamide | eNeuro

Probing Membrane Association of α-Synuclein Domains with VDAC Nanopore Reveals Unexpected Binding Pattern | Scientific Reports
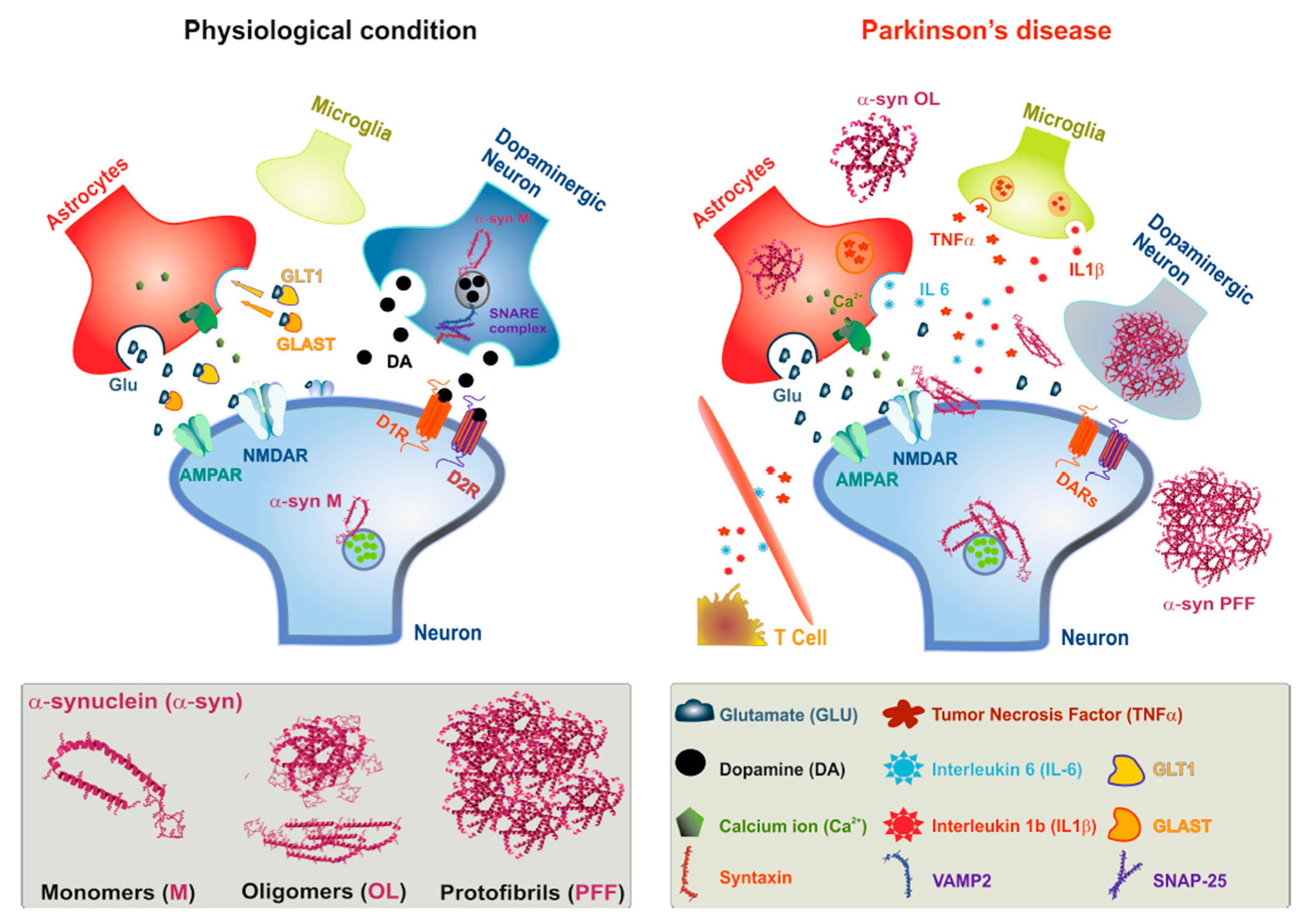
IJMS | Free Full-Text | Alpha-Synuclein as a Prominent Actor in the Inflammatory Synaptopathy of Parkinson's Disease | HTML

A Pathologic Cascade Leading to Synaptic Dysfunction in α-Synuclein-Induced Neurodegeneration | Journal of Neuroscience
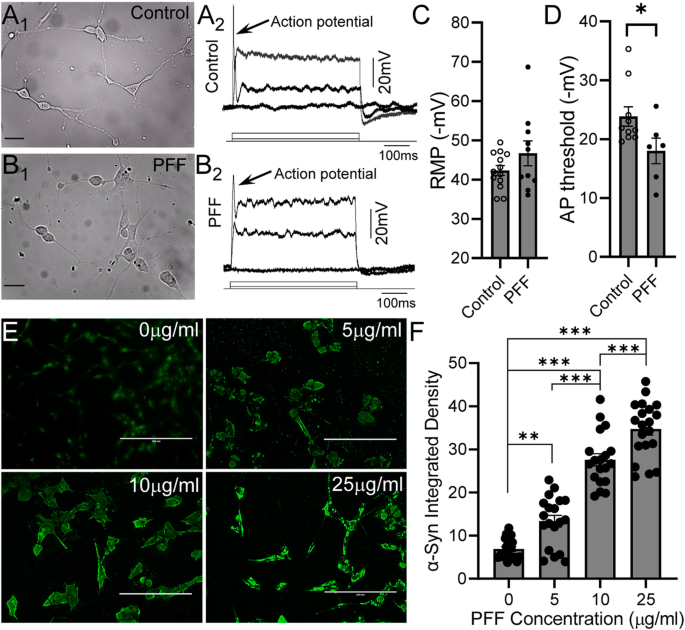
Alleviating toxic α-Synuclein accumulation by membrane depolarization: evidence from an in vitro model of Parkinson's disease | Molecular Brain | Full Text
Single-Channel Electrophysiology Reveals a Distinct and Uniform Pore Complex Formed by α-Synuclein Oligomers in Lipid Membranes
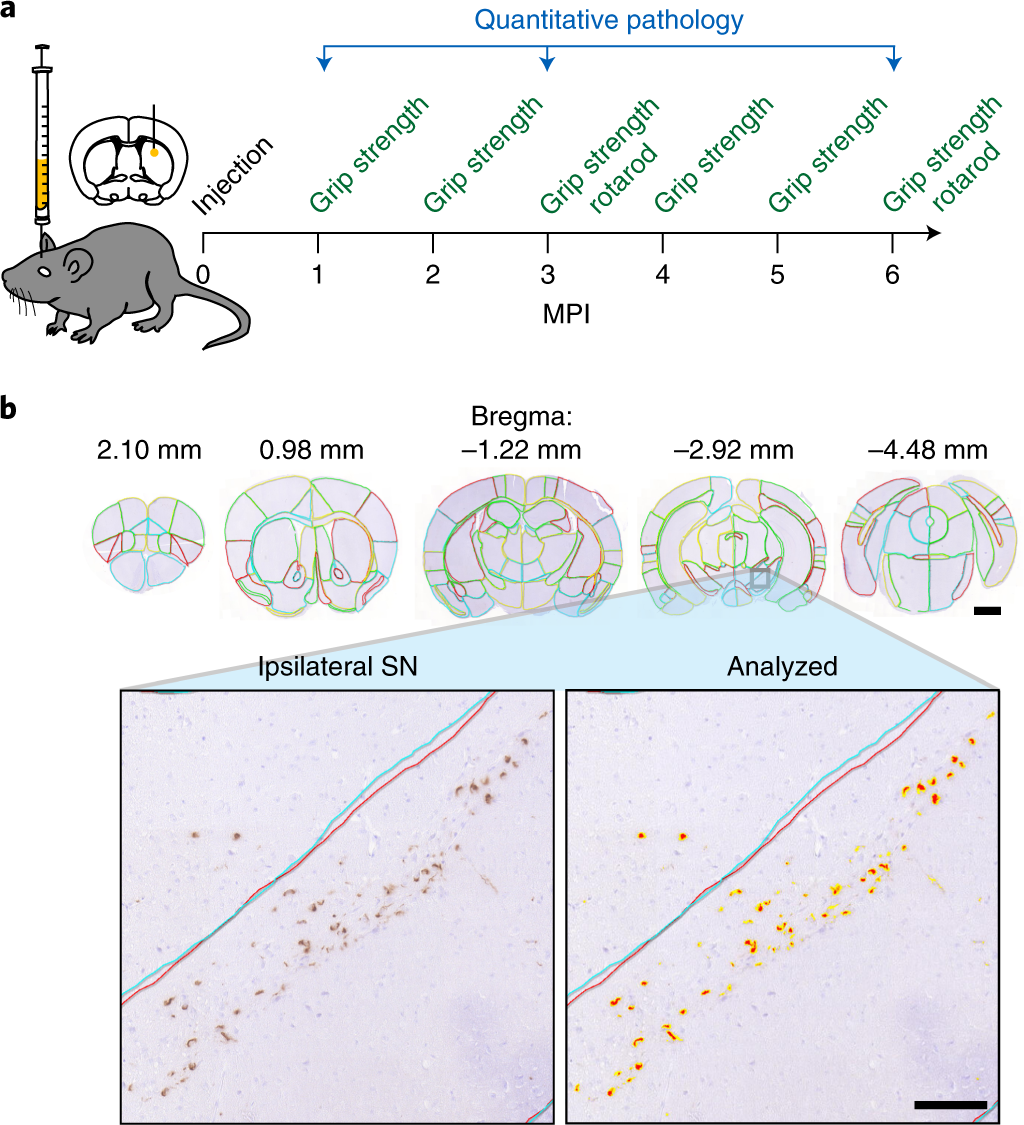
Spread of α-synuclein pathology through the brain connectome is modulated by selective vulnerability and predicted by network analysis | Nature Neuroscience

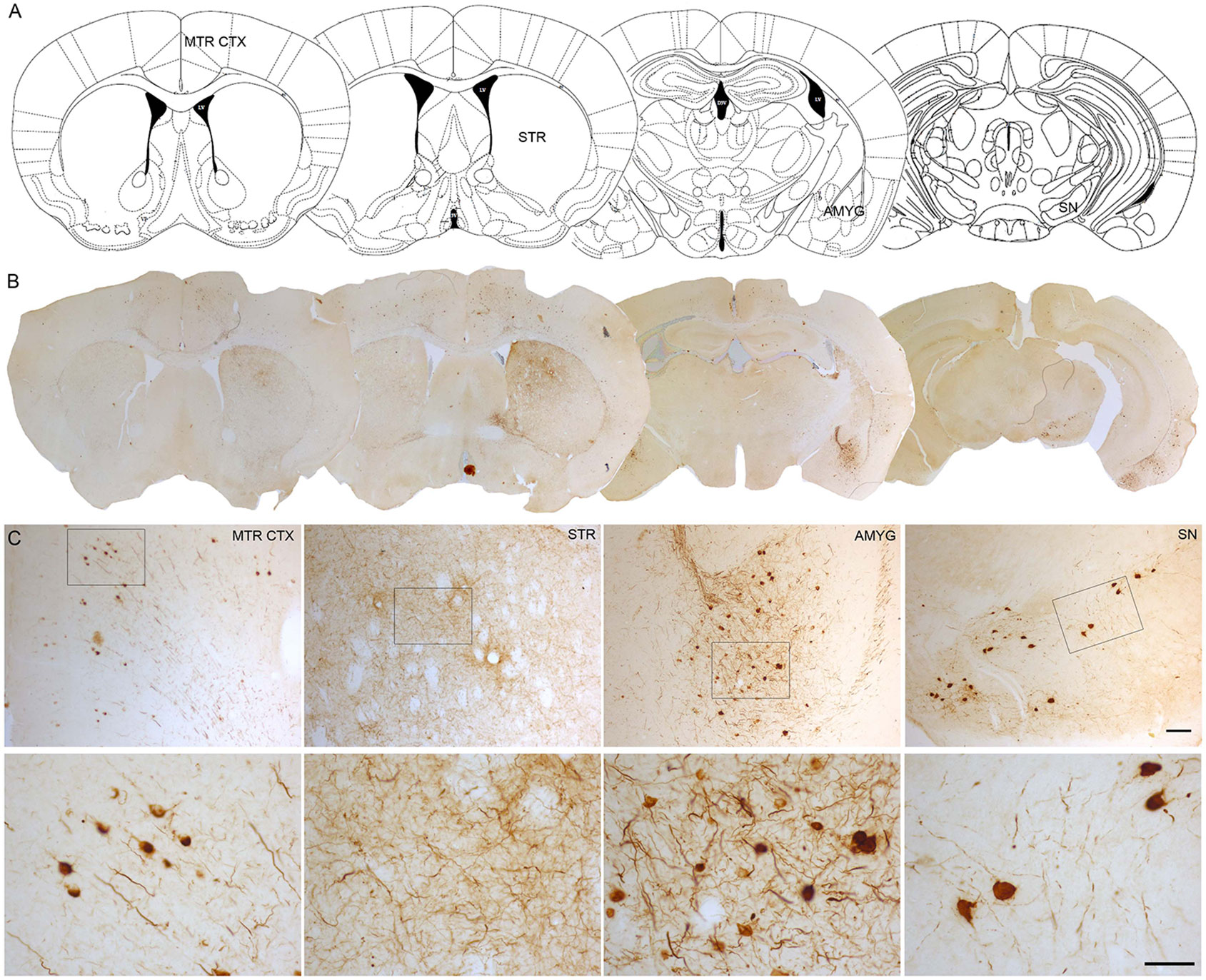
Characterization of SNpc intraneuronal α-Synuclein accumulations. (a)... | Download Scientific Diagram

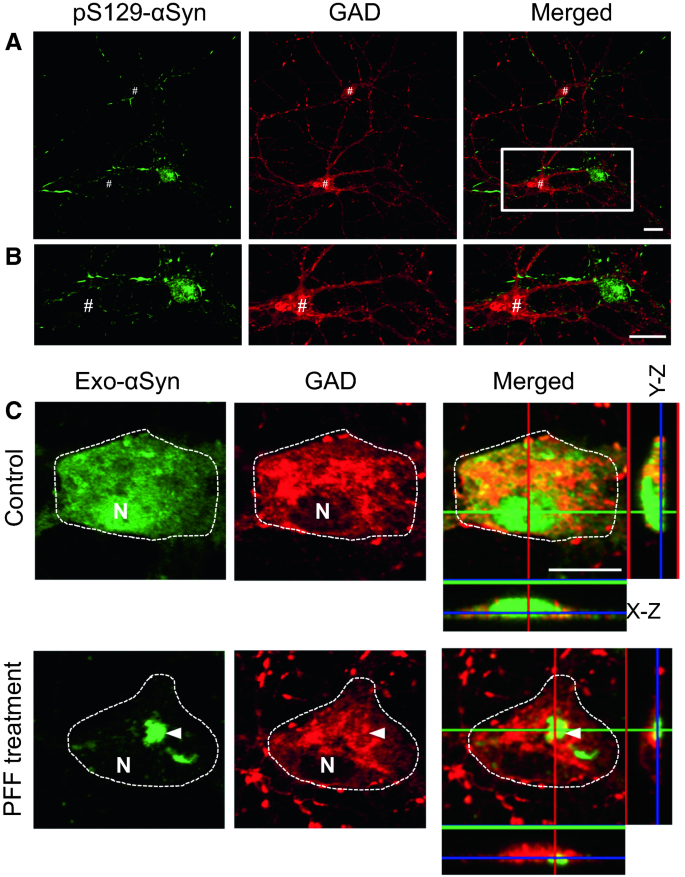
Exogenous Alpha-Synuclein Alters Pre- and Post-Synaptic Activity by Fragmenting Lipid Rafts - EBioMedicine

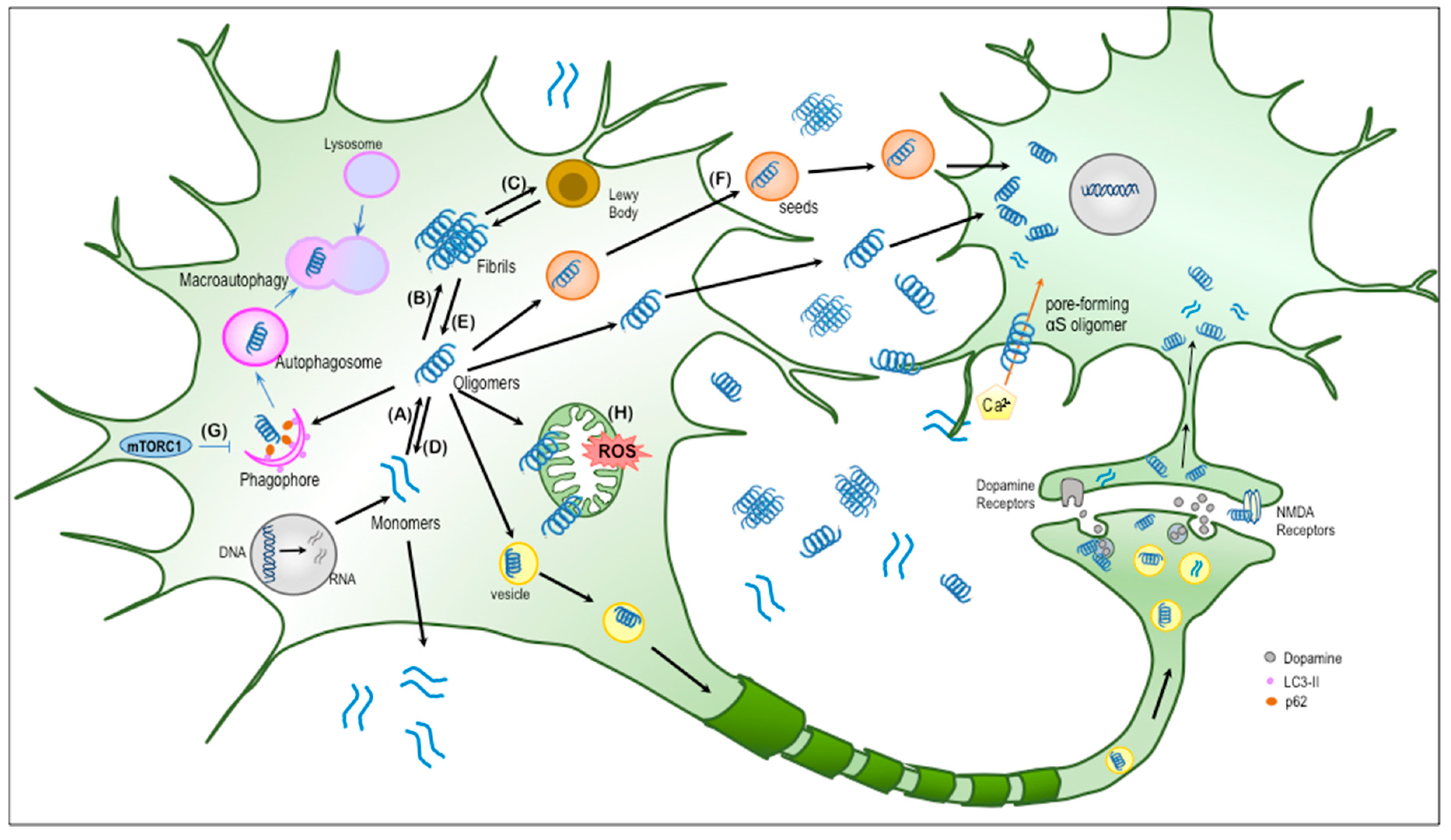
Alpha-synuclein at the intracellular and the extracellular side: functional and dysfunctional implications
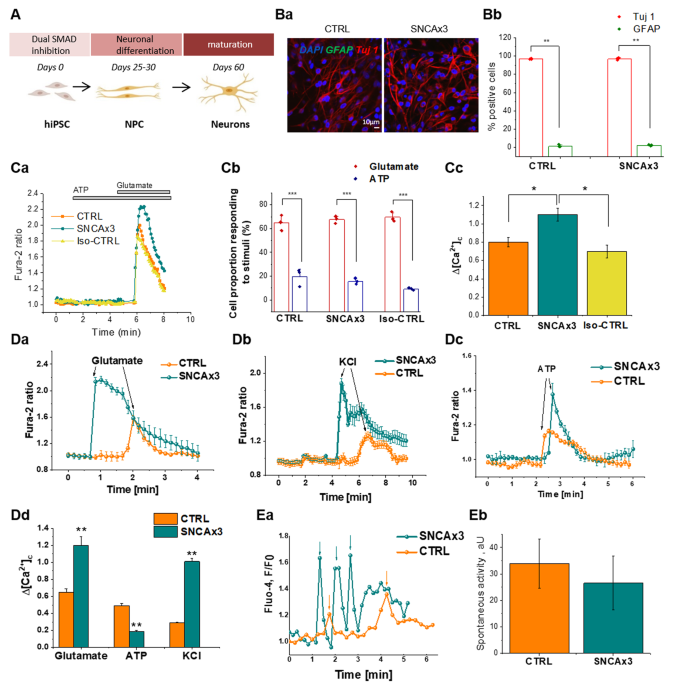
Alpha synuclein aggregation drives ferroptosis: an interplay of iron, calcium and lipid peroxidation | Cell Death & Differentiation
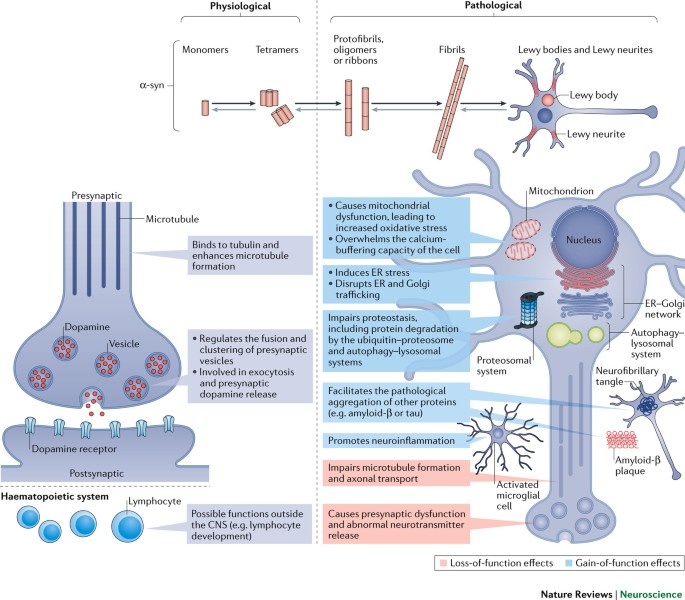
Animal models of α-synucleinopathy for Parkinson disease drug development | Nature Reviews Neuroscience
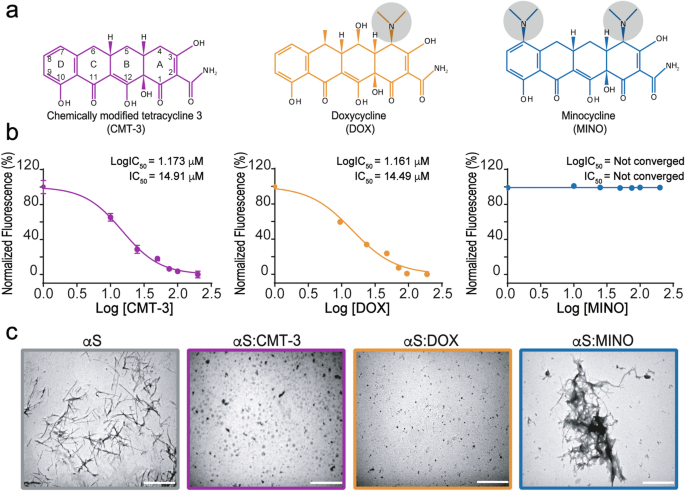
CMT-3 targets different α-synuclein aggregates mitigating their toxic and inflammogenic effects | Scientific Reports

α-Synuclein plasma membrane localization correlates with cellular phosphatidylinositol polyphosphate levels | eLife